
Now snap the two OxygenDB2s to the CarbonDB3s by the rectangular sockets. Snap this 3-atom section to the rest of the molecule by the two already installed bonds. Then snap the other CarbonDB3 between the two NItrogenSP21s by the round sockets (the NitrogenSP21s are symmetrical, but they look best if the face on the build platform is outside). Then snap a CarbonDB3 to the top bond of the Carbon56PH3 by the round socket (the rectangular socket should be on the outside) and put a bond in the other round socket. 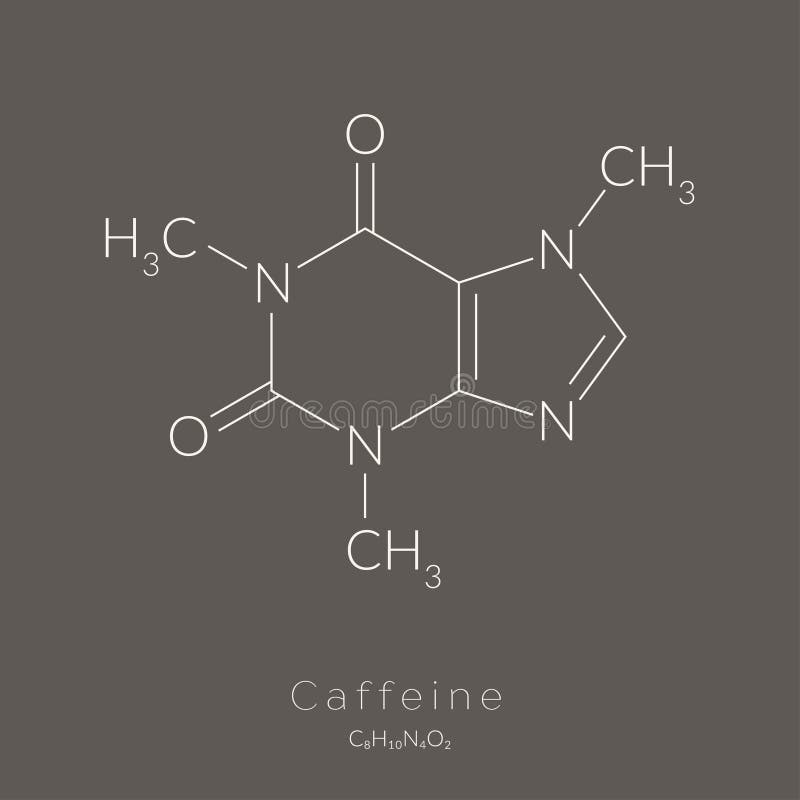
Put bonds in the free sockets of the Carbon56PH3. Then snap the three atoms to the Carbon56PH3 on the side with the shallower angle (you may have to flex the three-atom section a little). Connect the Nitrogen5PH1 and the black and blue Carbon5PH1s by the rectangular sockets. After the first end is in you should be able to snap on the second atom pretty easily. I've made STL files with all the atoms of each color if that helps. Lets get started.ġ0 Hydrogen1 in white (or another color of your choosing)ġ Carbon5PH1 in blue (this one will be nitrogen) And once you're done with that, you can also make theophylline (present in tea) and theobromine (present in chocolate). Caffeine is an interesting molecule chemically because it is made up of a 5-membered ring which is aromatic (a chemspeak term for a 5 or 6-membered ring which is planar and particularly stable) and a 6-membered ring which is almost but not quite aromatic (I'm not sure if this is more like almost dead or almost pregnant). Here, in one place is all you need to make everyone's favorite molecule, caffeine.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
